- An antigen is a substance or an element that has the ability to induce an immune response. Antigens are foreign substances and they are known to provoke the immune system to produce proteins known as antibodies, which act upon these antigens to eliminate them from the host.
- Antigens are foreign, which means that they are introduced into the body through certain mechanisms such as pathogenic infections, carcinogenic elements, induced immune reactions, etc.
- As foreign elements, antigens can be proteins, or lipopolysaccharide, or glycoproteins, or nucleoproteins.
- The human body’s immune system is designed to recognize and differentiate between self and non-self elements using an elaborate recognition mechanism. On recognizing non-self foreign elements, it then induces immune mechanisms to eliminate the antigens.
- This defines an antigen sometimes as an immunogen which is a substance that has the ability to induce immunogenicity i.e the ability to induce an immune response.
- Additionally, some antigens are self-induced antigens, therefore, the immune system reacts accordingly to eliminate both self and non-self antigens.
- Most antigens are large, complex molecules with a molecular weight generally greater than about 10,000. The ability of a molecule to function as an antigen depends on its size and structural complexity.
- However, some antigens are not large enough or complex enough to solely induce an immune response. Some are very small and they must be coupled with a larger carrier molecule in order to be recognized and an immune response induced against them. These small antigens are known as hapten.
- Haptens must be bound to a carrier protein for antibodies to be provoked against them and for the body to induce a T-cell immune response.
- The conjugation of a hapten to a carrier protein makes the hapten immunogenic, which means it can be recognized, processed, and presented to specific T-cells for elimination.
- When immune response can be elicited producing the lymphocytes (B- cells and T-cells), the response can be against the hapten or the carrier protein.
- Recognition of antigens by the immune responses is also determined by molecules found on the surface of the antigens known as antigen determinants or epitopes.
- Epitopes are regions in antigens that allow it to bind to the antigen-binding sites on antibodies or to the T-cell receptors. These epitopes or antigen determinants normally project from the antigen making it possible to bind readily to antibodies that already possess the sites for the antigen.
- The number of epitopes found on the antigen determines the number of binding antibodies. This number of epitopes is known as valence.
Interesting Science Videos
Epitope, Immunogen, hapten, and Tolerogen
- The antigenic determinants are discretely recognized by somatically generated B-cells and T-cell receptors. These cells have unique sets of receptors that recognize different epitopes, and they recognize them differently as well.
- For example, the B-cells recognize specific epitopes whether as part of the antigen (surface-bound) or as free-soluble molecules or as degraded/proteolytic fragments, while the T-cells only binds to epitopes that are on small fragments bound or affixed to specialized host cell surface molecules that display them such as macrophage cells.
Depending on the immune response the antigens trigger, epitopes can be classified into three:
Immunogen
- Immunogens possess epitopes that can induce an immune response and equally be the target of an immune response. As the body continuously reacts to the immunogen, an epitope-specific immune response is induced with increased intensity in its mechanism.
- However, the epitopes on antigens can bind to soluble or cell-surface receptors but not all antigens possess epitopes and therefore, not all antigens are immunogens.
- This means that antigens are molecules that have the ability to be recognized by the immune system, while an immunogen is a substance that evokes a specific immune response.
- Therefore, all immunogens are antigens but not all antigens are immunogens.
- Some non-immunogenic molecules can be bound to immunogens as carriers to help elicit an immune response. these non-immunogenic molecules are what we refer to as haptens.
- In this case, immunogens act as carriers.
Hapten
As defined above, haptens are small non-immunogenic antigens that can not solely be recognized by the immune receptors nor induce an immune response, and therefore it must be conjugated with a carrier protein, to enable its recognition. This means that happens are antigens but they are not immunogens. However, when it is bound to an immunogen (carrier) an immune response can be induced against the hapten and the epitopes found on the immunogen carrier.
Tolerogens
During immune responses, the body must develop tolerance to self-molecules first to prevent immune responses against self. This enables the immune system to subsequently recognize non-self antigens as foreign. After antigen elimination, the body must develop a long-term tolerance either after infection or by the administration of vaccines. This is where tolerogens develop their effect, whereby they induce adaptive immune unresponsiveness. So, unlike immunogens, exposure to tolerogens results in a diminished immune response rather than enhance it. Therefore, a tolerogen is a foreign antigen that induces a suppressed immune response or to produce an immune tolerance.
Properties of Antigens
Antigens possess a wide variety of features that largely determine the immunogenicity of these antigens. They include:
- Foreignness: This is a factor that confers the body the ability to recognize and distinguish between self and non-self molecules. Self molecules are not immunogenic and the molecules that are recognized as nonself.
- Size: Immunogenic molecules are large and complex, with a molecular weight of 10,000. Weak immunogens are weakly immunogenic while very small ones such as amino acids are non-immunogenic. The haptens become immunogenic when bound to a carrier protein.
- Structural complexity: Antigens require a certain amount of chemical complexity and a structure that has numerous, diverse antigenic determinants (epitopes) in order to induce an immune response than small simple peptides that contain a single of a few epitopes. For example, amino acids are homopolymers with one or two epitopes hence they are less immunogenic, unlike larger proteins or lipopolysaccharide or glycolipids which are heteropolymers with several epitopes, and containing two or more different amino acids, hence they are stronger immunogens.
- Chemical complexity: An immunogenic molecule has to be enzymatically cleavable by phagocytes. For example, the L-amino acid-containing polypeptides are very good and strong immunogens while the O-amino acid-containing polypeptides are poor immunogens because the proteolytic enzymes are able to cleave the L-amino acids only. Several molecules are immunogens including carbohydrates, steroids, and lipids, haptens, and amino acids are completely non-immunogenic.
- The genetic constitution of the host: Two strains of the same species of animals can respond differently to the same antigen. This is because of the difference in the composition of genes that are involved in the immune response, such as MHC alleles are normally different.
- Dosage, route, and timing of antigen administration: the degree of the immune response depends highly on the quantity of antigen administered therefore the immune response optimizes by redefining the dosage, route of administration, and the time of administration. Immunogenicity can be enhanced by combining it with an adjuvant (a substance that stimulates the immune response by facilitating the delivery of antigens to the Antigen-Presenting Cells (APCs)).
Types of Antigens
There are two main divisions of antigens that the body encounters;
Heteroantigens (Foreign antigens)
These are antigens that have an origin that is outside of the body. For example, viruses, bacteria, and parasites protozoans), the substances they produce, animal bites such as bee stings, snake venom, some foods like proteins (eggs, nuts) and components of serum and the red blood cells from other individuals (transfusion) are very typical sources of foreign antigens.
Autoantigens (Self-antigens)
These are antigens that originate from within the body. The body’s immunity has the ability to distinguish between self and non-self, but persons that have developed autoimmune disorders have a defected immune system where the normal body substances provoke an immune response against themselves leading to the generation of autoantibodies that attacks itself. Autoimmune disorders such as systemic lupus erythematosus, sclerosis, rheumatoid arthritis, etc are very common autoimmune disorders, due to self-destruction of the immune system.
Based on the above classification, Heteroantigens can still be classified into two groups. This is based on the type of microbial infection that attacks the body. Classification of antigens is normally based on the source of the antigen and the immune response they elicit on interaction with the host immune system.
Exogenous antigen
- These are antigens that gain entry into the body from the outside environment through ingestion, inhalation, or injection by the mechanisms of endocytosis or phagocytosis.
- Normally the reaction of the body’s immune system to exogenous antigens is by driving the endocytosed or phagocytosed antigens to antigen-presenting cells (APCs), such as macrophages, dendritic cells, where they are degraded into peptide fragments within the endocytic processing pathway.
- The antigen-presenting cells present the peptides to the T-helper cells known as the CD4+ using the Major Histocompatibility Complex (MHC) class II. The expression of class II MHC molecules is limited to antigen-presenting cells, and therefore, the presentation of exogenous peptide–class II MHC complexes is limited to these cells.
- Some antigens start out as exogenous and later become endogenous (for example, intracellular viruses). Intracellular antigens can be returned to circulation upon the destruction of the infected cell.
Endogenous antigen
- These are antigens that are produced within the host cell itself, as a result of normal cell metabolism or due to viral or intracellular bacterial infections or unique proteins synthesized by cancerous cells.
- The endogenous antigens are degraded into peptide fragments and presented on the cell surface of class I MHC molecules within the endoplasmic reticulum forming an endogenous peptide- class I MHC complex.
- This peptide-class I MHC complex is then transported to the cell membrane, activating the cytotoxic T-cell (CD8+).
- When the CD8+ recognizes them, they attack and kill all the cells that are displaying the endogenous antigen peptides-class I MHC complexes.
- The mechanism of the cytotoxic T cells involves the secretion of toxins that cause the lysis or the apoptosis of cells presented with the endogenous antigen fragments. However, to prevent the cytotoxic T-cells from damaging and killing cells because they present self-proteins, the cytotoxic self-reactive T cells are deleted by negative selection, which is a tolerance mechanism of the immune system.
- Endogenous antigens include xenogenic (heterologous), autologous, and idiotypic/allogenic (homologous) antigens.
- Xenogenic antigens
- Autologous antigens
- Allogenic or idiotypic or homologous antigens
Besides the above classification, there are other types of antigens, classified based on their sources. They include:
Superantigen
- Some antigens provoke drastic immune responses, and these are known as superantigens.
- They are normally bacterial proteins that stimulate T-cells to proliferate and interact with the Major Histocompatibility class II complex products, via the antigen-presenting cells and T-cell receptors.
- For example, staphylococcal enterotoxins that cause toxic shock syndrome is a superantigen.
- Superantigens cause symptoms by stimulating the release of large quantities of cytokines from T-cells. They are associated with chronic diseases such as rheumatic fever, arthritis, atopic dermatitis, psoriasis, and Kawasaki syndrome.
Tumor antigens
- These are antigenic substances that are produced by tumor cells, triggering immune responses by the body. They are normally presented by MHC class I or MHC class II on the surface of tumor cells.
- There are two types of tumor antigens i.e Tumor-specific antigens (TSA) and Tumor-associated antigens (TAA).
- Tumor-specific antigens (TSA) are found on the surfaces of the tumor cells and not on any other cells and they are a result of tumor-specific mutations while Tumor-associated antigens (TAA) are presented on tumor cells and some normal cells.
- Tumor cells when recognized by cytotoxic-T cells can be able to be destroyed.
- Some tumor antigens can appear on tumor surfaces as mutated receptors and they get recognized by B-cells.
- Human tumors that do not have viral origins, the tumor-specific DNA alterations form novel peptides (neopeptides).
Viral antigens
- These are antigens that are protein encoded by viral genomes.
- These antigens have multiple antigenicities which are protein in nature, they are species-specific, and they are closely associated with the viral particles.
- These antigens are specified by the viral genome that can be detected by the immunological response.
Neoantigens
- These are antigens that are absent from the normal human genome and they are used in tumor control because the quality of the T-cell pool available for these antigens is not affected by central T-cell tolerance.
- Viral-associated tumors like cervical cancer and other cancers, viral epitopes that are derived from viral open reading frames contribute to a pool of neoantigens.
- Neoantigen technology is a recent discovery, and it can be directly detected and quantified using MANA-SRM, a technique that was developed by the molecular diagnostic company, Complete Omics Inc., in collaboration with John Hopkins University School of Medicine.
Antigenic specificity
- Antigenic specificity allows the immune system to differentiate antigens. Antibodies are designed to recognize and differentiate between two protein molecules that have a difference in only a single amino acid. The capability of the immune system to generate tremendous diversities in recognizing molecules give it the ability to recognize billions of unique structures found on foreign antigens.
- After the immune system has recognized and produced a response to the antigen, the immune system then generates an immunological memory, which generates a response when it encounters the antigen the second time, with a heightened immune reactivity. immunological memory confers a long-term or life-long immunity to many infectious agents after an initial encounter.
- Lastly, the immune system reacts to foreign antigens, which means that it is able to recognize and differentiate between the self and non-self antigens. This unique property of the immune system defines the responses elicited on interaction with a foreign antigen in the system. However, the immune system can also react to self-antigens with fatal outcomes.
Antigen Recognition
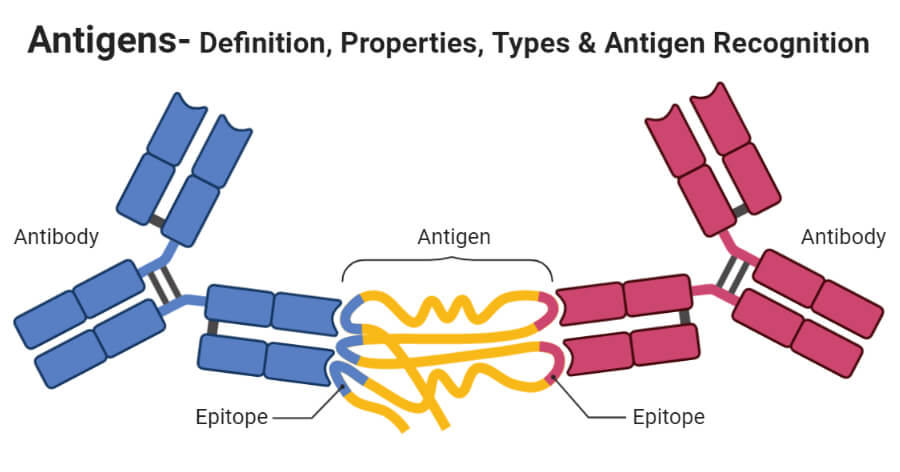
- Antigen recognition is achieved when the immune system is able to distinguish nonself molecules and molecules from self, which are part of the body. This is the role of the immune system in identifying and neutralizing foreign antigens as a body defense mechanism.
- As we discussed above, antigens have molecules on or in them known as antigen determinants or epitopes. The epitopes are immunologically active regions of immunogens that whose role is to bind antigenic-specific membrane receptors found on lymphocytes or secreted antibodies.
- B-cells and T-cells recognize different epitopes on the same antigenic molecule.
- The lymphocytes can interact with complex antigens on several levels of the antigen structure. An epitope on a protein antigen has primary, secondary, tertiary, and quaternary structures of the protein, while in the polysaccharide antigens, the epitopes may be branch chains.
- The difference in the way the lymphocytes recognize antigens is by the mechanism played by the B and T-lymphocytes.
- The B-cells recognize soluble antigens when they bind to their membrane-bound antibodies, and since they can bind antigens that are freely found in solutions, they are able to recognize epitopes that are highly accessible sites on the exposed surface of the immunogens.
- The T-cells recognize peptides that are bound to MHC molecules on the surface of antigen-presenting cells and altered self-cells.
B-cells Epitope recognition
- B-cell epitopes are determined by antigen-binding sites found on the antibody molecules displayed by B-cells. The antibodies bind to the epitopes by short, weak non-covalent interactions. To form a strong bond, the epitope and the antibody’s binding site must-have complementary shapes placing the interacting groups near each other, therefore, the epitopes should have some restriction properties. The size of the epitopes to be recognized by B-cells should and can not be larger than the size of the antibody’s binding site.
- The B-cell epitopes found on native proteins are made up of hydrophilic amino acids on the protein surface that are easily accessible to the membrane-bound or free antibody. Accessibility enables fast and easy binding to antibodies, hence these antigens possess protrusions on its surface which are recognized as epitopes.
- The B-cell epitopes are located in flexible regions of immunogens and they display site mobility. Site mobility increases or maximizes complementarity with the antibody’s binding site allowing the antibody to bind to the epitope. If the sites are rigid, binding is ineffective, however, it possesses high affinity as compared to binding to flexible epitopes.
- Complex protein antigens such as globular proteins have multiple overlapping B-cell epitopes, hence can be recognized by a wide range of different antibodies.
- The B-cell epitopes can have sequential or nonsequential amino acids. The sequential amino acid residues are found along the polypeptide chain and the nonsequential amino acid residues are from segments of chains that are brought together by the folded conformation of an antigen.
- The receptors that bind epitopes on the B-cell are known as the B-cell receptors or Fc receptors (FcR) and they are found on the Fc portion of the antibodies IgA, lgG, or lgM.
- Fc receptors (FcRs) are expressed on the surfaces of phagocytic cells. Phagocytic cells recognize and bind epitope-engaged antibodies, which leads to the phagocytosis of the epitope-antibody-FcR complex. An antibody that does not bind to an epitope continues to circulate in the body.
T-cell Epitopes Recognition (T-cell receptors)
- T-cells recognize antigens that have been processed to antigenic peptides that are bound to the Major Histocompatibility Complex (MHC) molecule and do not bind denatured native proteins or free circulating proteins.
- The antigenic peptides that are recognized by T-cells form trimolecular complexes with a T-cell receptor and an MHC molecule. Therefore, the T-cell epitopes must have the ability to interact with both T-cell receptors and an MHC molecule.
- The binding of the MHC molecule to the antigenic peptides is not specific in its interaction between the antibody and its epitope, but instead, an MHC molecule selectively binds several different peptides.
- The antigenic peptides are generated by antigen processing, which interacts specifically with the MHC molecule. the endogenous and exogenous antigens are processes by different intracellular pathways in that, the endogenous antigens are processes into peptides within the cytoplasm while the exogenous antigens are processes by the endocytic pathway.
- The epitopes that are recognized by the T-cells are internal. The T-cells have a tendency to recognize internal peptides that are exposed by processing within the antigen-presenting cells or altered self-cells.
- T-cell Receptors are associated with the cluster of differentiation (CD) complex of the transmembrane surface molecules, and specifically CD3. Other clusters of differentiation are also associated with the coreceptors of the T-cell receptors.
Pattern recognition receptors (PRRs)
- The Pattern recognition receptors are designed to recognize and to bind only nonself structures that are abundantly circulating in the microbial world but are not expresses in the normal host cells.
- The PRR’s pattern structure is encoded in the genome hence they are transmitted across generations and expresses in every individual within the species in essentially identical forms.
- The PRRs are effective in the innate immune responses, identifying structures that are associated with microbes and not by the host cells.
- A typical example of the Pattern Recognition Receptors is the Toll-like receptors which are found on the membranes of various cell types. Other PRRs are soluble and are found in the cytoplasm of the body fluids.
The cluster of Differentiation (CD)
- The cluster of differentiation (CD) specific cells bearing membrane proteins (antigens) that are used in quantifying the reaction of lymphocytes produce in response to various diseases. They are functional surface proteins or cell receptors that can be measured in situ and from peripheral blood, biopsy, or body fluids. They are used to differentiate between leukocyte subpopulations.
- There are over 350 CDs the have been identified.
- They have both biological and diagnostic functions.
- The serum of a normal human has a low concentration of these proteins but when the body is activated due to disease, their concentrations increase drastically, and therefore, monitoring of cD levels helps in disease management.
For example;
- CD54, also known as intercellular adhesion molecule-1 (ICAM-1), is a protein that is an important early marker of immune activation and response. It is a ligand for the lymphocyte function-associated antigen-1 (LFA-1) found on lymphocytes, monocytes, and neutrophils. The CD54/LFA-1 interaction is important in T-cell–antigen-specific responses and lymphocyte emigration into inflammatory sites. CD54 also is found on mucosal epithelial cells and erythrocytes. Recent evidence suggests CD54 is the cellular receptor for the human rhinovirus and Plasmodium falciparum-infected erythrocytes. The circulation of CD54 relates to the progress and prognosis of skin cancer metastatic melanoma,
- CD23 which is a B-cell growth factor, is a protein found on the surface of IgM-bearing B cells, eosinophils, macrophages, and platelets. Soluble CD23 is a B-cell growth factor and upregulates IgE synthesis in conjunction with interleukin-4 released from T cells. When its levels are elevated, it is associated with chronic lymphocytic leukemia.
- CD35 is a glycoprotein that is present on erythrocytes, Poly MorphoNuclear cells (PMNs), monocytes, all B-cells, and some T cells, mast cells, and in the soluble form in the plasma. It is the receptor for complement C3b/C4b. The functions of CD35 include binding of immune complexes on erythrocytes that bear C3b or C4b and promote their clearance from the circulation, CD35 augments the maturation of B-cells into antibody-secreting cells, aiding leukocytes in binding and phagocytosing C3b- or C4bcoated objects. CD35 is also a potential agent for suppression of complement-dependent tissue injury in autoimmune inflammatory disease.
- The CD8 molecule is a polypeptide found on the surface of cytotoxic T cells. It is a molecule that participates in the interaction of class I
MHC found on target cells. Elevated CD8 is found in children with lymphoid malignancies and in HIV-infected individuals - CD4 is a glycoprotein that is found on T-cells associated with helper/inducer regulatory functions and also reduces the degree on monocyte and macrophages. It is a cell adhesion molecule that has a great affinity for class II MHC.
- The CD2 is a glycoprotein present on T cells, thymocytes, and NK cells. It functions as an intercellular adhesion molecule and binds
to the human leukocyte function-associated antigen-3 or LFA-3 (CD58). CD2 may be involved in the attachment of TH cells to antigen-presenting cells and cytotoxic T cells to target cells, and in other cellular interactions. It also is a signal-transducing molecule and may help stimulate T cells to proliferate and secrete cytokines.
Applications of Antigens
- Antigens are used for diagnosis of infections in that, identification of the presence of an antigen is an individual showing symptoms of a disease. When the individual recovers, the antigen disappears.
- Detection of autoimmune disorders is enabled by the detection of self-antigens
- Antigens are also used in the preparation of vaccines, which induces an immune reaction and creates a lasting memory against the antigen, hence the use of vaccines to prevent infections.
References
- Immunology by Kuby, 5th Edition
- Microbiology by Prescott, 5th Edition
- Lippincott’s Illustration Review in Immunology
- Comprehensive immunology: Antigens and Immunogens by Frederic C. McDuffie
- Medical Microbiology by Jawertz, 23rd Edition
- Immunobiology: The Immune System in Health and Disease. 5th edition: Chapter 3, Antigen Recognition by B-cell and T-cell Receptors
- http://resources.schoolscience.co.uk/unilever/16-18/proteins/Protch7pg1.html
- https://microbiologyonline.org/index.php/about-microbiology/microbes-and-the-human-body/antibody-antigen-complex
- https://www.sigmaaldrich.com/technical-documents/articles/biology/antigens-epitopes-antibodies.html
- https://www.technologynetworks.com/immunology/articles/antigen-vs-antibody-what-are-the-differences-293550
- https://en.wikipedia.org/wiki/Antigen
- https://www.britannica.com/science/antigen
- https://www.webmd.com/diabetes/qa/what-are-antigens
- https://bio.libretexts.org/Bookshelves/Microbiology/Book%3A_Microbiology_(Kaiser)/Unit_6%3A_Adaptive_Immunity/12%3A_Introduction_to_Adaptive_Immunity/12.2%3A_Antigens_and_Epitopes
- https://www.news-medical.net/life-sciences/The-Characteristics-of-Antigens.aspx
- https://www.biologyonline.com/dictionary/tolerogen
- https://www.toleranzia.com/technology/tolerogen-platform/
- https://en.wikipedia.org/wiki/Tumor_antigen#:~:text=Tumor%20antigen%20is%20an%20antigenic,cancer%20immunology%20studies%20such%20topics.
- https://www.prospecbio.com/viral_antigens#:~:text=A%20viral%20antigen%20is%20a,influenza%2C%20hepatitis%2C%20%26%20AIDS.
- https://www.creative-diagnostics.com/Viral-Antigens.htm
- https://www.msdmanuals.com/professional/hematology-and-oncology/tumor-immunology/tumor-antigens
- https://en.wikipedia.org/wiki/Cluster_of_differentiation
- https://www.sinobiological.com/research/cd-antigens/what-is-cluster-of-differentiation

very nice notes…Thank you